 
In addition, some macromolecules and cell structures can act as heterogeneous crystallization nuclei, inducing the precipitation. This releasing of molecules increases pH and elevates the supersaturation, resulting in the precipitation of minerals. The precipitation of minerals by microorganisms is obtained by the modification of the local environment as a result of the metabolites release. The second is distinguished by massive intracellular and/or extracellular mineral formation commonly in the form of teeth, skeletons, shells, etc. The first takes place in various animals in a process where the organism produces an organic framework to introduce ions for further crystallization and growth mediated by an organic matrix. This mineral formation occurs through two different processes. Minerals precipitation by living organisms activity, so-called biomineralization, is a process that occurs from bacteria to chordates. MICP is proposed as a potentially safe and powerful procedure for efficient conservation of worldwide heritage structures. The treatment has revealed best results on porous media matrixes nevertheless, it can also be applied on soil, marble, concrete, clay, rocks, and limestone. This method has shown to be successful as a restoration, consolidation, and conservation tool for improvement of mechanical properties and prevention of unwanted gas and fluid migration from historical materials. In this work, some published variations of a novel and ecological surface treatment of heritage structures based on MICP are presented and compared. Historic buildings and artwork, especially those present in open sites, are susceptible to enhanced weathering resulting from environmental agents, interaction with physical-chemical pollutants, and living organisms, among others. The high concentration of carbonate and calcium ions on the bacterial surface, which serves as nucleation sites, promotes the calcium carbonate precipitation filling and binding deteriorated materials. The patina avoids further copper degradation below the oxidised base.Microbiologically induced carbonate precipitation (MICP) is a well-known biogeochemical process that allows the formation of calcium carbonate deposits in the extracellular environment. Thus, copper is regarded as an important corrosion resistant element. 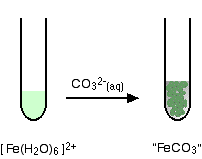
The patina serves as a shielding shield as opposed to other harmful oxidation processes and it does not induce any degradation in the metal. If the atmosphere contains high moisture content then this cycle would be easier. Similar to iron and aluminium, if exposed to air, the copper element undergoes the oxidation process. Note- Oxidation is a phenomenon that causes one element to lose electrons (and/or hydrogen) when interacting with another. Hence, the colour of copper carbonate is green. The copper powder reacts with oxygen, forming an opaque coating of copper oxide that tends to be green or bluish-grey in color. Oxidation of Copper and Changing of Color After some time copper becomes green due to its exposure to air.Ĭopper carbonate always stays as copper carbonate hydroxide in the atmosphere. Once copper is exposed to sunlight, the metal reacts to form a combination of copper carbonate and copper hydroxide with moisture and natural gases. 
Basic copper carbonate (the "copper carbonate" of commerce) is actually a copper carbonate hydroxide.Ĭopper metal turns gray in colour when exposed to dust due to corrosion. On the basis of its reaction with air present in the atmosphere and the chemical abundance of copper carbonate, we will find the colour of copper carbonate.Ĭopper carbonate is a rarely seen moisture-sensitive compound. Hint- In order to find the colour of copper carbonate, first we will see the basic state or chemical state in which copper carbonate stays.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
