
Please let us know how we can improve this web app. It is also sometimes called: Molecular Mass, Molecular Weight, Formula Mass, or Formula Weight. One atomic mass unit (u) is equal to 1/12 the mass of one atom of carbon-12. You can view more details on each measurement unit. We assume you are converting between grams Carbon and mole. Convert grams Carbon to moles - Conversion of Measurement Units. Related: Molecular weights of amino acids Molar mass is the mass (in atomic mass units) of one mole of a of a substance. Do a quick conversion: 1 grams Carbon 0.083259093974539 mole using the molecular weight calculator and the molar mass of C. Weights of atoms and isotopes are from NIST article. 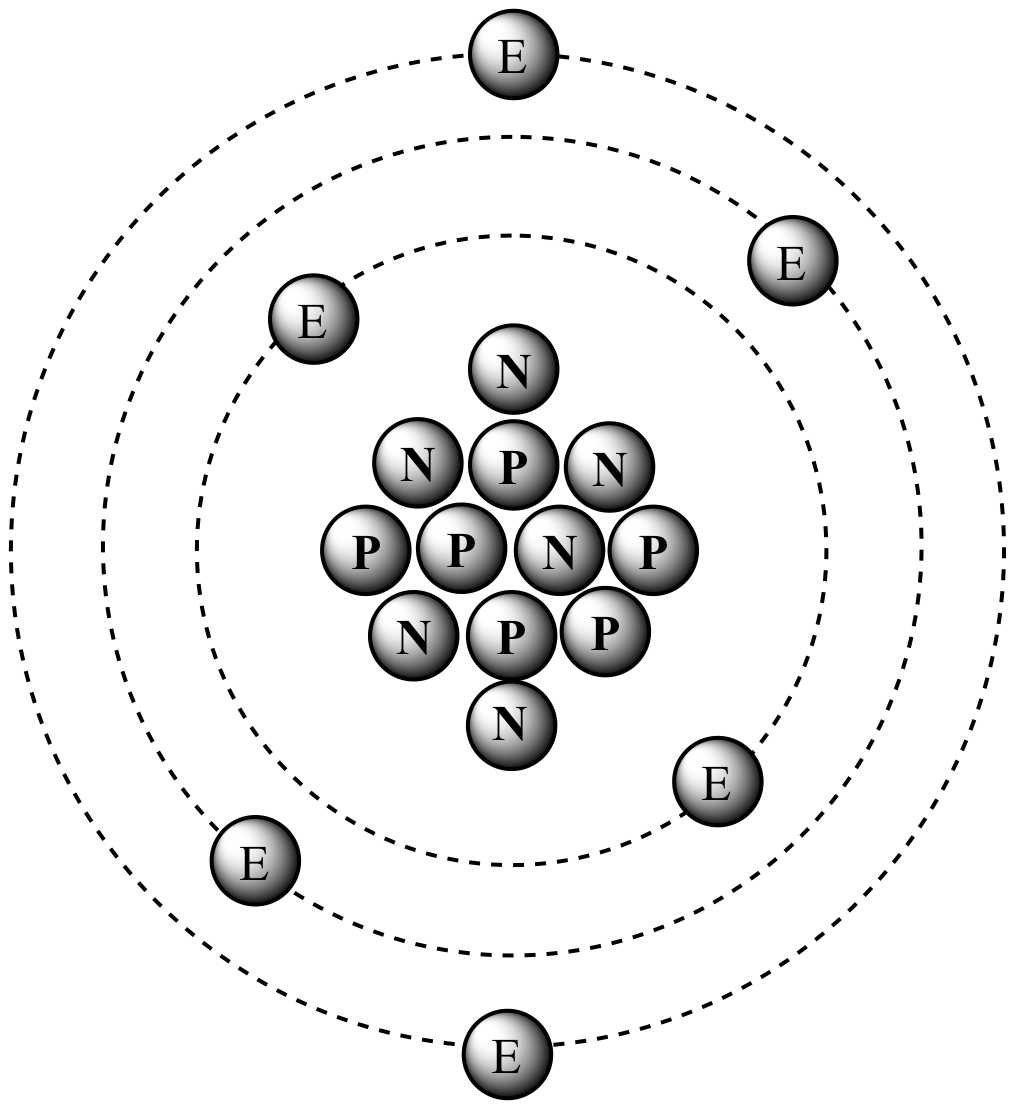
Molar mass ( molar weight) is the mass of one mole of a substance and is expressed in g/mol.(1 u is equal to 1/12 the mass of one atom of carbon-12) Molecular mass ( molecular weight) is the mass of one molecule of a substance and is expressed in the unified atomic mass units (u). Compound X has a molar mass of 153.03 g mol 1 and the following composition: element mass carbon 47.09 hydrogen 6.59 chlorine 46.33 Write the molecular formula of X. 
To calculate molecular weight of a chemical compound enter it's formula, specify its isotope mass number after each element in square brackets.Įxamples of molecular weight computations:ĭefinitions of molecular mass, molecular weight, molar mass and molar weight Molar mass calculator also displays common compound name, Hill formula, elemental composition, mass percent composition, atomic percent compositions and allows to convert from weight to number of moles and vice versa.Ĭomputing molecular weight (molecular mass)
0 Comments
Leave a Reply. |
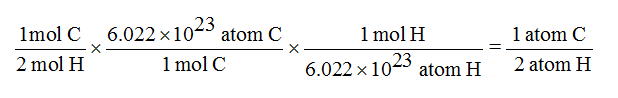
 RSS Feed
RSS Feed
